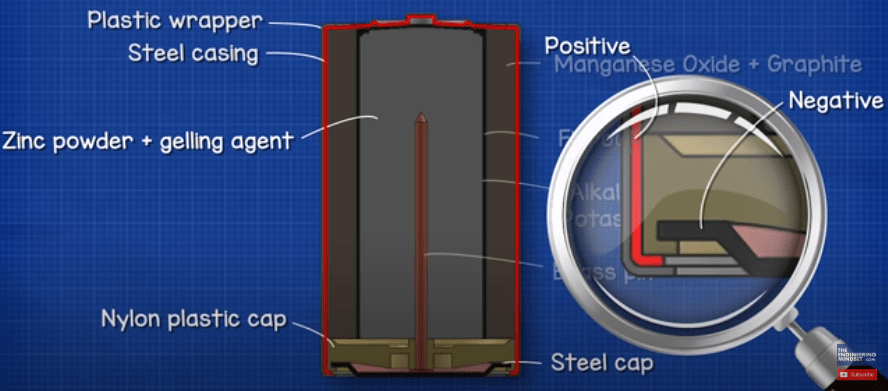
A typical 1.5V alkaline battery looks something like this, but the colours will vary by manufacturer. When we look at the battery, we usually have a plastic wrapper fitted tightly to the outside, this will insulate the battery but also tell us important information such as the capacity and voltage as well as which end is the positive and negative.
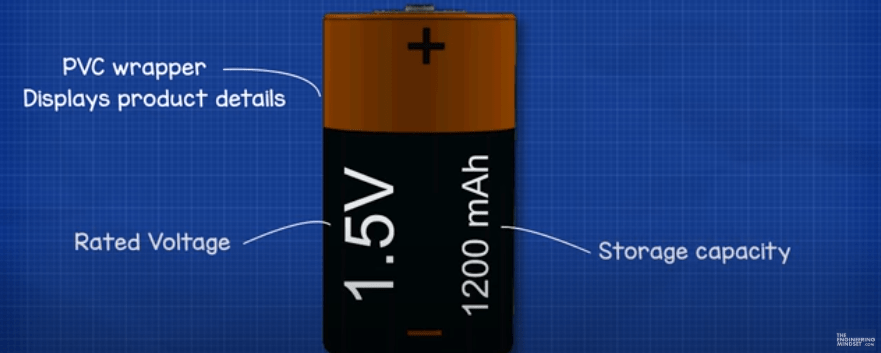
The positive end is known as a cathode and will have this extended surface which protrudes outwards.

The negative end will be flat, the negative end is known as the anode. These two terminals are electrically isolate from each other.

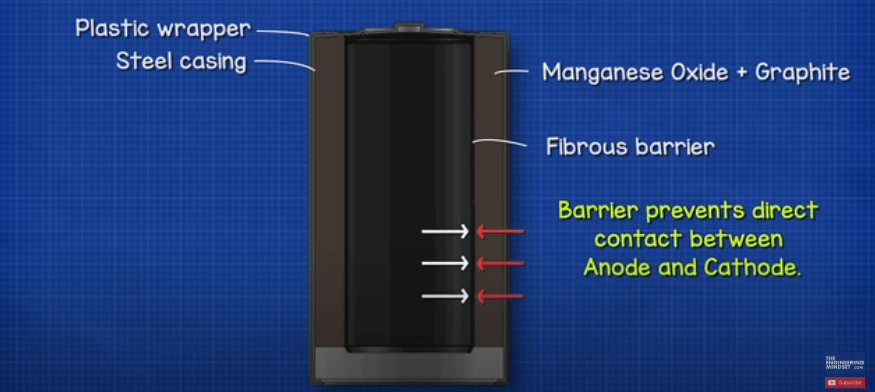
Under the wrapper we find the main casing, which is usually made from steel with a nickel plating. This holds all the internal components in place and stops them interacting with elements of the atmosphere such as air and water.

Within the casing we have multiple layers of different materials, these materials are specially selected because their chemical reactions create certain levels of voltages and current. The first layer is the cathode which is a mixture of manganese oxide (MnO2) and graphite. This material is in contact with the metal of the positive terminal. The graphite is added to improve the conductivity of the mixture and increases the energy density.

Next, we find a layer of porous material, typically a fibrous paper which forms a barrier. The barrier prevents the anode and cathode materials from having direct contact with each other, this helps the battery last longer when it’s not being used. If the barrier wasn’t there then the battery would short circuit. The microscopic holes within the material allows ion atoms to pass through it. We will look at that in more detail later in this article.
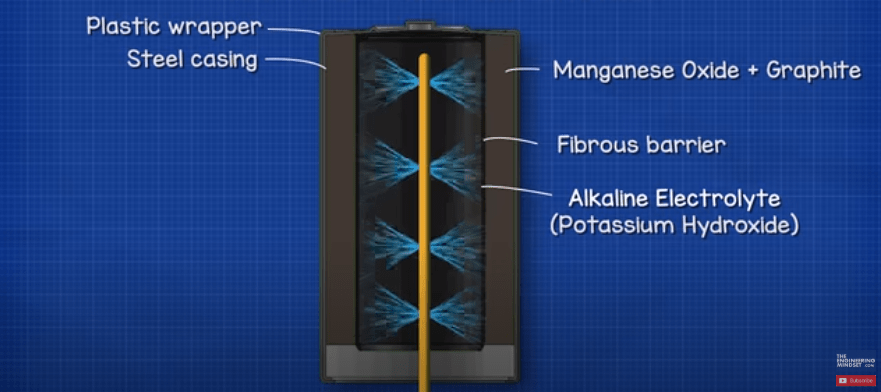
An electrolyte liquid of potassium hydroxide is then sprayed onto the seperator during the manufacturing process which soaks it and will be absorbed into the anode material. The electrolyte used is an alkaline which is why we refer to this type of battery as an alkaline battery.
On the other side of the barrier we have the anode which is a paste made from zinc powder (Zn) as well as a gelling agent. The gelling agent just keeps the zinc suspended so it doesn’t accumulate in one spot. The zinc is in a powder form to increase the surface area of the material which lowers the internal resistance and thus improves electron transfer.
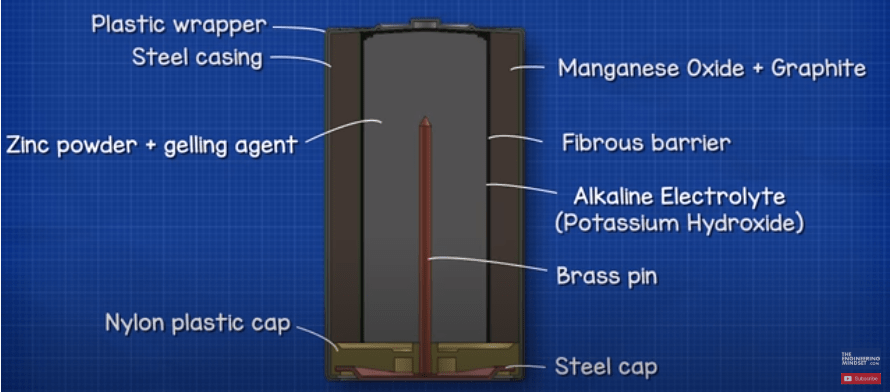
The steel capsule is sealed with a nylon plastic cap. A brass pin is then inserted into the zinc with a steel cap placed over this. This gives us the negative terminal. Notice that the positive and negative terminals are separated by the plastic cap. This ensures they are electrically isolated from each other, otherwise electrons could flow through the casing to reach the positive terminal and short circuit the battery.
Leave a Reply